|
The reactivities of the carbohydrate and aglycon partners can be finely-tuned by introducing different protecting groups. Overview of Glycosylation PrinciplesĬarbohydrate synthesis requires a crafty approach in selecting protecting groups, participating groups, promoter systems, glycosyl donor and selectively deprotected glycosyl acceptors in order to achieve stereo-controlled glycosylation in reasonable yields ( Figure 1A). One approach to conjugating carbohydrates to binding partners of interest (i.e., protein or lipid) in enantiomerically pure form is through the stereoselective installment of linkers with functionalizable handles, which can be elaborated to generate complex glycans and/or multivalent displays.ġ.2. In either case, achieving the desired stereochemical specificity and multiplicity of ligation products with high purity remains a challenge. Chemoenzymatic methods offer a complementary approach however, enzyme availability, substrate specificity and scalability can hinder product diversity. To address these limitations, synthetic platforms that afford large-scale production of pure and chemically defined glycoconjugates are under development. Common isolation techniques often require enzymatic digestion, detergent extraction and multiple purifications, which may degrade the sugars although recent methods to extract O-glycans using bleach hold promise for commercial use. Like most natural products, glycoconjugates are typically difficult to isolate and may only occur as heterogenic mixtures in scarce amounts. 
Understanding how structure gives rise to function is critical for the development of carbohydrate-based therapeutics and expedient access to synthetic materials is a significant challenge for researchers in this area. Deciphering the “sugar codes” created by specific sequences of oligosaccharides linked to lipid and protein anchors is an emerging area of glycomics, which like proteomics has the underlying goal of connecting chemical structures to biological functions. Other biomolecules such as glycolipids and glycosylphosphatidylinositols (GPI anchor) display hydrophilic carbohydrate moieties that participate in ligand-receptor binding, cell-to-cell interactions and pathogenic processes such as bacterial and viral infection as well as cancer metastasis. Glycosylation is a prominent form of post-translational modification occurring in a majority of eukaryotic proteins. With the launch of the Human Genomic Project in the beginning of this century, efforts have shifted toward understanding structure/function relationships of post-translational modifications of proteins. In contrast, carbohydrates have multiple similarly reactive hydroxyl groups that connect with varying region- and stereochemistries giving rise to a complex set of structures that have no corresponding genetic blueprint. DNA, RNA and the proteins they encode have relatively confined connectivity and predictable chemistries owing to the limited number of ways the nucleic acid and amino acid building blocks can be combined.  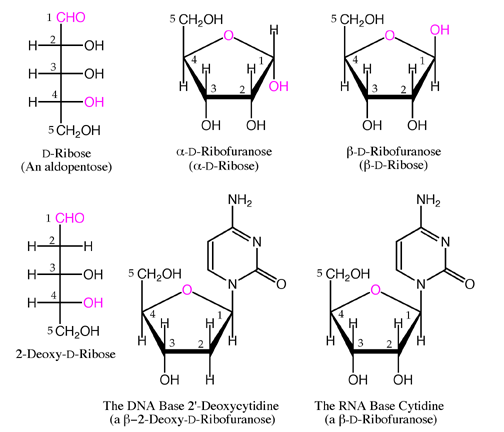
Nucleic acids, proteins and carbohydrates are three important classes of biomolecules.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
